The U.S. Food and Drug Administration has approved Tapentadol hydrochloride, an immediate-release oral tablet. This approval offers health care professionals an additional choice for relieving patients' moderate to severe acute pain.
The new drug, brand named Tapentadol ®, manufactured by Janssen Ortho, LLC, Gurabo, PR, is a centrally-acting synthetic analgesic that is made available in doses of 50 mg, 75 mg, or 100 mg.
Tapentadol was made to act in two ways, opioid (narcotic) and non-opioid. It affects the brain and body primarily by activating opioid receptors in the brain, spinal cord and gastrointestinal tract. Additionally, Tapentadol inhibits the reuptake of the brain chemical norepinephrine which is suspected to have an analgesic effect.
Acute pain accompanies many medical conditions and significantly interferes with the patients' every day's functioning and a quality of life.
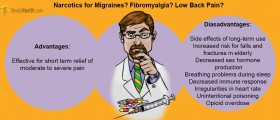
Opioids are a good choice for pain management but can, however, cause dependence, abuse, and addiction. This is why all patients treated with opioids require careful monitoring by their doctors to determine when their use is no longer needed and to check for signs of abuse and addiction.
Tapentadol ® most reported side effects include nausea, dizziness, vomiting, and sleepiness. The labeling for Tapentadol includes warnings about the risk of respiratory depression; addictive depressive effects on the central nervous system when taken with alcohol, other opioids, or illicit drugs; and abuse potential.
The new drug, brand named Tapentadol ®, manufactured by Janssen Ortho, LLC, Gurabo, PR, is a centrally-acting synthetic analgesic that is made available in doses of 50 mg, 75 mg, or 100 mg.
Tapentadol was made to act in two ways, opioid (narcotic) and non-opioid. It affects the brain and body primarily by activating opioid receptors in the brain, spinal cord and gastrointestinal tract. Additionally, Tapentadol inhibits the reuptake of the brain chemical norepinephrine which is suspected to have an analgesic effect.
Acute pain accompanies many medical conditions and significantly interferes with the patients' every day's functioning and a quality of life.
Opioids are a good choice for pain management but can, however, cause dependence, abuse, and addiction. This is why all patients treated with opioids require careful monitoring by their doctors to determine when their use is no longer needed and to check for signs of abuse and addiction.
Tapentadol ® most reported side effects include nausea, dizziness, vomiting, and sleepiness. The labeling for Tapentadol includes warnings about the risk of respiratory depression; addictive depressive effects on the central nervous system when taken with alcohol, other opioids, or illicit drugs; and abuse potential.
Loading...