Table of Contents
The Ebola virus infection is an easily transmitted, highly infectious, and very deadly disease that has claimed the lives of close to 2,000 people. The virus has killed fewer than 3,000 people since it was identified only about 40 years ago, but it has everybody increasingly concerned because it can kill up to 90% of people who are infected. In the past, Ebola infections were controlled by isolating the infected patients in small settlements. Recently, however, a large outbreak has affected more people from East Africa, including those from Liberia, Sierra Leone, Nigeria, and Guinea. Furthermore, scientists still have to find out how infection can be prevented and how affected individuals can be cured from the disease.
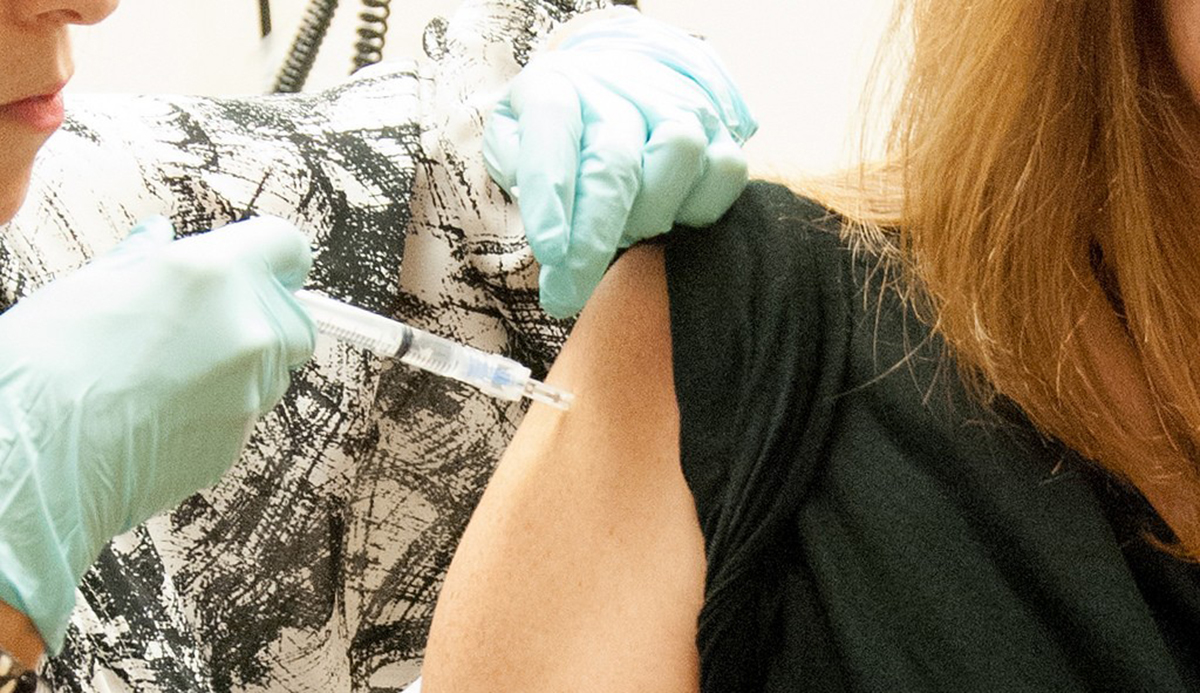
Ebola Vaccine: Still Experimental
To protect uninfected individuals from Ebola virus infection, experimental vaccines are now being developed and tested for safety and effectiveness. Vaccines are drugs that can stimulate the body's immune system to build a response against viruses.
Clinical trials for the vaccine will involve healthy adults who have not been infected with the virus and will determine if the vaccine induces an effective immune response and is safe to use. The World Health Organization (WHO) has declared the Ebola epidemic an international health emergency and clinical trials using the vaccines will soon be underway. This is to ensure that the vaccines will be available for use by 2015.
One vaccine developed by the pharmaceutical GlaxoSmithKline has shown promising results when used on monkeys, and will be tested on a few human subjects (phase 1) in clinical trials to be conducted in Bethesda, Maryland. Although it usually takes years for drugs like vaccines to get final approval for marketing from the FDA due to its strict standards, exceptions may be made, especially when faced with a deadly disease that can spread like wildfire. Other vaccines that are being developed include those from the NewLink Genetics Corp and the Public Health Agency of Canada, and those supported by the National Institutes of Health (NIH).
One problem with developing a vaccine is marketing it, since Ebola infection tends to be sporadic and unpredictable, and therefore, doctors will have difficulty recommending it to populations who may not even be at risk. However, with the urgency of the current situation, the NIH has announced that clinical trials will be beginning soon.
See Also: US Citizen In Ghana Does Not Have Ebola
Antivirus Drug Looks Promising
Meanwhile, an experimental antivirus drug designed to treat already infected individuals has also been developed and is now being tested in monkeys. This drug is called ZMapp (by Mapp Biopharmaceutical Inc.) and it consists of three monoclonal antibodies that can neutralize the virus by binding to their protein and disrupting replication and/or destroying them. Results of studies using infected monkeys showed that treatment within 24 hours led to full recovery, but if treatment was delayed for several days, only half of the infected monkeys survived. No formal studies of human subjects have been done. Other experimental drugs that are still being developed include TMK-Ebola, favipiravir, AVI-7537, BCX-4430, estrogen receptor modulators, and ST-383.
- The Telegraph. Ebola vaccine won't be ready until 2015, says WHO. http://www.telegraph.co.uk/news/worldnews/africaandindianocean/liberia/11024010/Ebola-vaccine-wont-be-ready-until-2015-says-WHO.html
- WebMD. Experimental Ebola Serum Grown in Tobacco Leaves.http://www.webmd.com/news/20140804/ebola-virus-vaccine
- ABCNews. How an Ebola Vaccine Could Change the Next Deadly Outbreak.http://abcnews.go.com/Health/ebola-vaccine-change-deadly-outbreak/story?id=24825398
- CDC. Questions and Answers on Experimental Treatments and Vaccines for Ebola.http://www.cdc.gov/vhf/ebola/outbreaks/guinea/qa-experimental-treatments.html
- NY Times. Ebola Vaccine Possible, but Many Doubts Persist.http://www.nytimes.com/2014/08/02/health/effort-to-develop-ebola-vaccine-to-test-on-humans-is-put-on-fast-track.html
- MedicineNet. Ebola Vaccine: Is It Safe?http://www.medicinenet.com/ebola_vaccine_is_it_safe/views.htm
- CDC. Ebola (Ebola Virus Disease) Prevention.http://www.cdc.gov/vhf/ebola/prevention/index.html
- Photo courtesy of NIAID by Flickr : www.flickr.com/photos/niaid/15137998885
- Photo courtesy of NIAID by Flickr : www.flickr.com/photos/niaid/15138207362
- www.telegraph.co.uk
- www.webmd.com
- abcnews.go.com
- www.cdc.gov
- www.nytimes.com
- medicinenet.com


Your thoughts on this