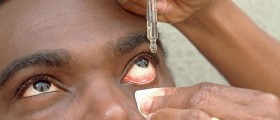
The FDA has approved a new clinical trial of retinal implants that helped blind people get their visions back. These retinal implants are called Argus II Retinal Prosthesis System and have been developed by the from the Doheny Eye Institute, Keck School of Medicine, University of Southern California, San Francisco.
The scientists tested their initial 16 electrodes implants on blind people who lost their visions from eyes diseases such as pigmentosa or macular degeneration. These people had damaged photoreceptor cells in their retina and the implants were inserted into their eyes. Six patients who were blind were able to see light, detect movement, and identify some shapes and objects.
What they are planning to do next is test their new implants with 60 electrodes and provide the patients with better eyesight.
These retinal implants work by communicating with an external camera and computer. The patients also receive a pair of glasses with a video camera. Visual data from the camera is processed by a computer, the processed data is then sent to the implant wirelessly. The electrodes from the implant turn the data into electrical impulses that are then being sent into the brain, so that it could interpret the visual data.
For the time being, there are around 25 million people worldwide are blind due to pigmentosa or macular degeneration and the number is expected to rise. The trials will include patients over 50 who were able to see well before they started suffering from the disease. This new technology will not provide them with the eyesight of a person with healthy vision but the difference will certainly be huge.
The scientists tested their initial 16 electrodes implants on blind people who lost their visions from eyes diseases such as pigmentosa or macular degeneration. These people had damaged photoreceptor cells in their retina and the implants were inserted into their eyes. Six patients who were blind were able to see light, detect movement, and identify some shapes and objects.
What they are planning to do next is test their new implants with 60 electrodes and provide the patients with better eyesight.
These retinal implants work by communicating with an external camera and computer. The patients also receive a pair of glasses with a video camera. Visual data from the camera is processed by a computer, the processed data is then sent to the implant wirelessly. The electrodes from the implant turn the data into electrical impulses that are then being sent into the brain, so that it could interpret the visual data.
For the time being, there are around 25 million people worldwide are blind due to pigmentosa or macular degeneration and the number is expected to rise. The trials will include patients over 50 who were able to see well before they started suffering from the disease. This new technology will not provide them with the eyesight of a person with healthy vision but the difference will certainly be huge.
Loading...