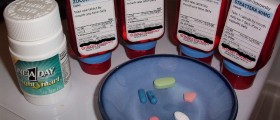
The U.S. Food and Drug Administration have finally approved marketing of several generic versions of drugs that treat HIV. Their approval was blocked earlier due to patent and marketing exclusivity restrictions and the drugs were not available in the US.
This move helps reduce the expense for people in the United States being treated for the disease. AZT, an anti-retroviral drug that is also known as Zidovudine, helps prevent the AIDS virus from reproducing in the body. It is often used in combination with other medications to treat an HIV infection.
It is very important to mention that this generic version of the drug has been approved for children’s use as well.
“The Emergency Plan for AIDS Relief, which President Bush first announced in his 2003 State of the Union Address, is currently providing $15 billion to fight the HIV/AIDS pandemic over five years, with a special focus on 15 of the hardest-hit countries. The President's Emergency Plan is designed to prevent seven million new HIV infections, treat at least two million HIV-infected people, and care for ten million HIV-affected individuals, AIDS orphans and vulnerable children.”
This move helps reduce the expense for people in the United States being treated for the disease. AZT, an anti-retroviral drug that is also known as Zidovudine, helps prevent the AIDS virus from reproducing in the body. It is often used in combination with other medications to treat an HIV infection.
It is very important to mention that this generic version of the drug has been approved for children’s use as well.
“The Emergency Plan for AIDS Relief, which President Bush first announced in his 2003 State of the Union Address, is currently providing $15 billion to fight the HIV/AIDS pandemic over five years, with a special focus on 15 of the hardest-hit countries. The President's Emergency Plan is designed to prevent seven million new HIV infections, treat at least two million HIV-infected people, and care for ten million HIV-affected individuals, AIDS orphans and vulnerable children.”
Loading...